Carbon Storage
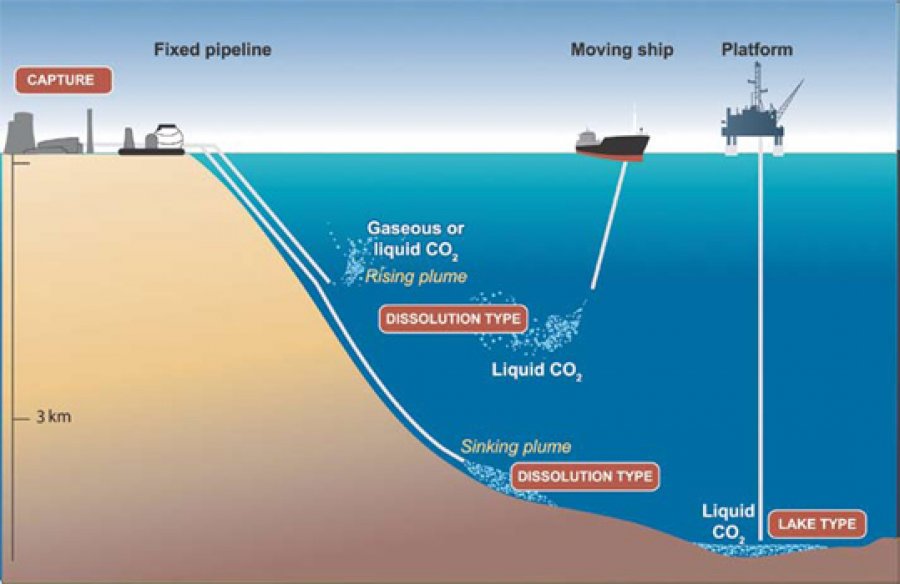
Carbon storage relates to storing carbon dioxide in man made or naturally occurring structures such as old oil wells, or deep into the sea.
Carbon Dioxide Scrubbers

These scrubbers capture carbon dioxide from the air, which would be particularly useful where the gas was produced in particularly high volumes.
Biochar

Biochar is produced by burning Biomass without oxygen. This creates a very stable substance, that locks in CO2 and can be used to fertilise soil.
Bio CCS Algal Synthesis
Bio CCS Algal Synthesis attempts to capture carbon dioxide using micro algae, and instead of storing it, it aims to transform it into useful products.
Artificial Photosynthesis

Artificial Photosynthesis is a way of mimicking the natural process that plants use to create energy from sunlight, CO2 and water.